NfrA
Revision as of 15:10, 17 April 2014 by 134.76.70.252 (talk)
- Description: Spx-dependent FMN-containing NADPH-linked nitro/flavin reductase, stress protein
| Gene name | nfrA |
| Synonyms | ywcG, ipa-43d |
| Essential | no |
| Product | FMN-containing NADPH-linked nitro/flavin reductase |
| Function | unknown |
| Gene expression levels in SubtiExpress: nfrA | |
| Metabolic function and regulation of this protein in SubtiPathways: nfrA | |
| MW, pI | 28 kDa, 5.732 |
| Gene length, protein length | 747 bp, 249 aa |
| Immediate neighbours | ywcH, rodA |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 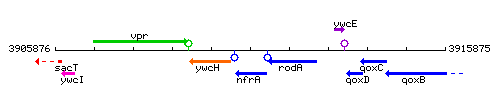
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed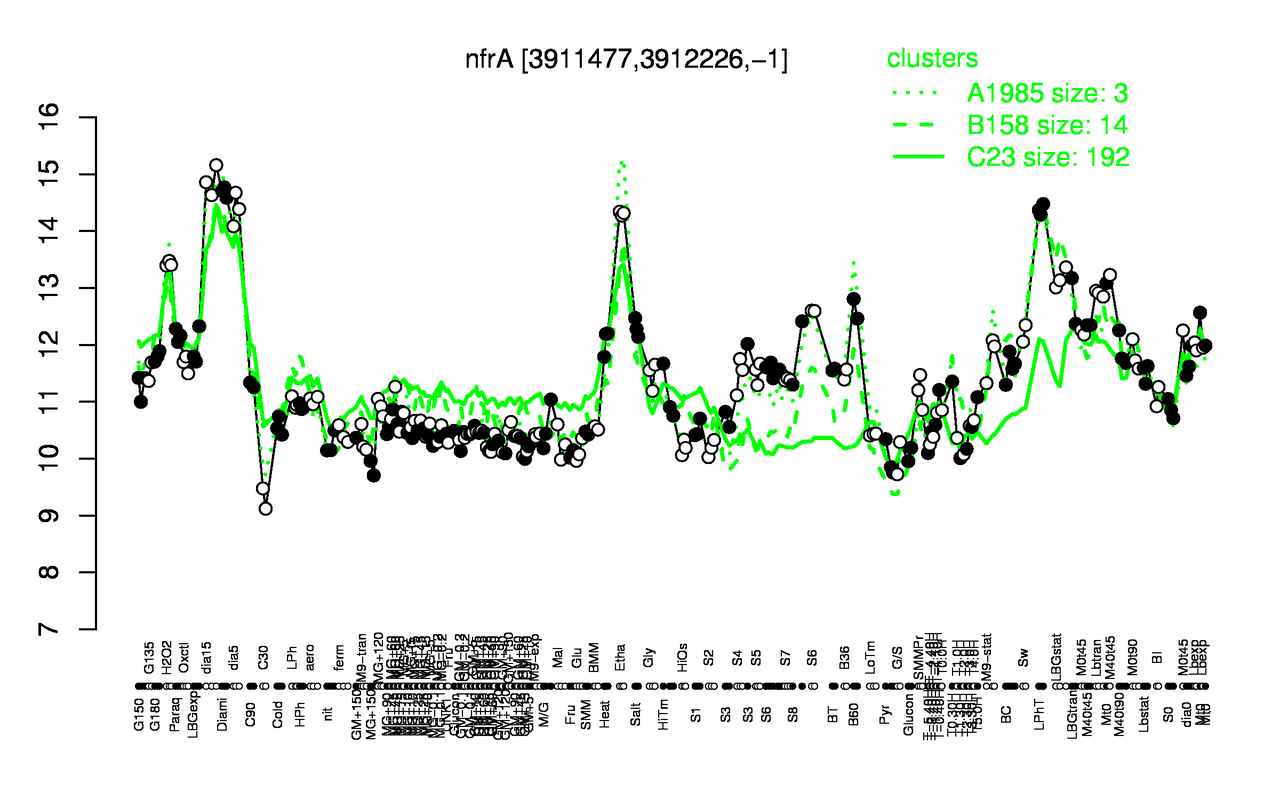
| |
Contents
Categories containing this gene/protein
electron transport/ other, resistance against oxidative and electrophile stress
This gene is a member of the following regulons
SigD regulon, Spo0A regulon, Spx regulon
The gene
Basic information
- Locus tag: BSU38110
Phenotypes of a mutant
Database entries
- BsubCyc: BSU38110
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: possesses an NADH oxidase activity that leads to high concentrations of oxygen peroxide and an NAD(+) degrading activity leading to free nicotinamide PubMed
- Protein family: flavin oxidoreductase frp family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
Database entries
- BsubCyc: BSU38110
- UniProt: P39605
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Additional information:
- number of protein molecules per cell (minimal medium with glucose and ammonium): 222 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, exponential phase): 616 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, early stationary phase after glucose exhaustion): 560 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, late stationary phase after glucose exhaustion): 960 PubMed
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References