Difference between revisions of "SigA"
(→References) |
(→Extended information on the protein) |
||
| Line 100: | Line 100: | ||
** [[BsrB]]-[[SigA]]-[[RNA polymerase]] | ** [[BsrB]]-[[SigA]]-[[RNA polymerase]] | ||
| − | * '''[[Localization]]:''' | + | * '''[[Localization]]:''' |
| + | ** cytoplasm {{PubMed|25313396}} | ||
=== Database entries === | === Database entries === | ||
Revision as of 17:40, 27 October 2014
- Description: RNA polymerase major sigma factor SigA
| Gene name | sigA |
| Synonyms | rpoD, crsA |
| Essential | yes PubMed |
| Product | RNA polymerase major sigma factor SigA |
| Function | transcription |
| Gene expression levels in SubtiExpress: sigA | |
| Interactions involving this protein in SubtInteract: SigA | |
| MW, pI | 42 kDa, 4.634 |
| Gene length, protein length | 1113 bp, 371 aa |
| Immediate neighbours | cccA, dnaG |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 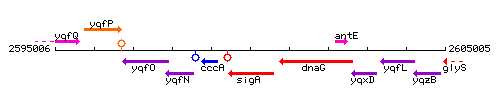
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed
| |
Contents
Categories containing this gene/protein
transcription, sigma factors and their control, essential genes, membrane proteins
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU25200
Phenotypes of a mutant
essential PubMed
Database entries
- BsubCyc: BSU25200
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family: sigma-70 factor family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Localization:
- cytoplasm PubMed
Database entries
- BsubCyc: BSU25200
- Structure:
- UniProt: P06224
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Regulation:
- Regulatory mechanism:
- Additional information:
- An antisense RNA is predicted for sigA PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium): 802 PubMed
- number of protein molecules per cell (complex medium with amino acids, without glucose): 1811 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, exponential phase): 1136 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, early stationary phase after glucose exhaustion): 2062 PubMed
- number of protein molecules per cell (minimal medium with glucose and ammonium, late stationary phase after glucose exhaustion): 1407 PubMed
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
- Charles Moran, Emory University, NC, USA homepage
Your additional remarks
References
Reviews
Benedikt Steuten, Sabine Schneider, Rolf Wagner
6S RNA: recent answers--future questions.
Mol Microbiol: 2014, 91(4);641-8
[PubMed:24308327]
[WorldCat.org]
[DOI]
(I p)
Lakshminarayan M Iyer, L Aravind
Insights from the architecture of the bacterial transcription apparatus.
J Struct Biol: 2012, 179(3);299-319
[PubMed:22210308]
[WorldCat.org]
[DOI]
(I p)
Original publications