HprK
| Gene name | hprK |
| Synonyms | ptsK, yvoB |
| Essential | no |
| Product | HPr kinase/ phosphorylase |
| Function | carbon catabolite repression, phosphorylation of HPr and Crh proteins at Ser46 |
| Gene expression levels in SubtiExpress: hprK | |
| Interactions involving this protein in SubtInteract: HprK | |
| Metabolic function and regulation of this protein in SubtiPathways: hprK | |
| MW, pI | 34 kDa, 4.906 |
| Gene length, protein length | 930 bp, 310 aa |
| Immediate neighbours | lgt, nagA |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 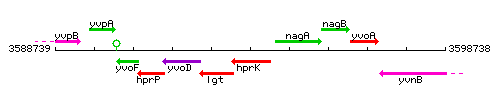
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed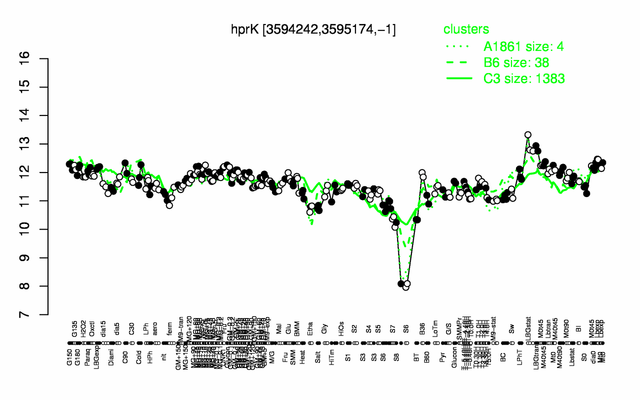
| |
Contents
Categories containing this gene/protein
protein modification, transcription factors and their control
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU35000
Phenotypes of a mutant
no carbon catabolite repression
Database entries
- DBTBS entry: no entry
- SubtiList entry: [1]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: ATP + HPr = ADP + P-Ser-HPr (according to Swiss-Prot)
- Protein family: HPrK/P family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
Database entries
- Structure: 1KKM (complex of Lactobacillus casei HprK with B. subtilis HPr-Ser-P), 1KKL (complex of Lactobacillus casei HprK with B. subtilis HPr)
- UniProt: O34483
- KEGG entry: [2]
- E.C. number:
Additional information
Expression and regulation
- Regulation:
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant: GP202 (spc) PubMed, GP858 (aphA3) PubMed, GP82 (cat) PubMed, all available in Jörg Stülke's lab
- Expression vector:
- for expression/ purification from B. subtilis with N-terminal Strep-tag, for SPINE, in pGP380: pGP642, available in Jörg Stülke's lab
- for expression/ purification of mutant HprK-G158A from B. subtilis with N-terminal Strep-tag, for SPINE, in pGP380: pGP650, available in Jörg Stülke's lab
- for expression/ purification from E. coli with N-terminal His-tag, in pWH844: pGP205, available in Jörg Stülke's lab
- for expression, purification of the N-terminal in E. coli with N-terminal His-tag, in pWH844: pGP218, available in Jörg Stülke's lab
- lacZ fusion: pGP201 (in pAC5, PubMed) available in Stülke lab, pGP202 (in pAC6, PubMed) available in Jörg Stülke's lab
- GFP fusion:
- two-hybrid system:
- Antibody: available in Jörg Stülke's lab PubMed
Labs working on this gene/protein
Josef Deutscher, Paris-Grignon, France
Jörg Stülke, University of Göttingen, Germany Homepage
Wolfgang Hillen, Erlangen University, Germany Homepage
Anne Galinier, University of Marseille, France
Your additional remarks
References
Reviews
General Analysis, Physiology
Structural Analysis of HPrK
Enzymatic Properties, Mutation Analysis
HprK as a Target For Antimicrobial Compounds