Difference between revisions of "Sandbox"
| Line 1: | Line 1: | ||
| − | * '''Description:''' | + | * '''Description:''' pyruvate dehydrogenase (dihydrolipoamide acetyltransferase E2 subunit) <br/><br/> |
{| align="right" border="1" cellpadding="2" | {| align="right" border="1" cellpadding="2" | ||
|- | |- | ||
|style="background:#ABCDEF;" align="center"|'''Gene name''' | |style="background:#ABCDEF;" align="center"|'''Gene name''' | ||
| − | |'' | + | |''pdhC'' |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"| '''Synonyms''' || '' | + | |style="background:#ABCDEF;" align="center"| '''Synonyms''' || '' '' |
|- | |- | ||
|style="background:#ABCDEF;" align="center"| '''Essential''' || no | |style="background:#ABCDEF;" align="center"| '''Essential''' || no | ||
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"| '''Product''' || | + | |style="background:#ABCDEF;" align="center"| '''Product''' || pyruvate dehydrogenase (dihydrolipoamide acetyltransferase E2 subunit) |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"|'''Function''' || | + | |style="background:#ABCDEF;" align="center"|'''Function''' || links glycolysis and TCA cycle |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"| '''MW, pI''' || | + | |style="background:#ABCDEF;" align="center"| '''MW, pI''' || 47 kDa, 4.855 |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"| '''Gene length, protein length''' || | + | |style="background:#ABCDEF;" align="center"| '''Gene length, protein length''' || 1326 bp, 442 aa |
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"|'''Immediate neighbours''' || ''[[ | + | |style="background:#ABCDEF;" align="center"|'''Immediate neighbours''' || ''[[pdhB]]'', ''[[pdhD]]'' |
|- | |- | ||
| − | |style="background:#FAF8CC;" align="center"|'''[http://subtiwiki.uni-goettingen.de/ | + | |style="background:#FAF8CC;" align="center"|'''[http://subtiwiki.uni-goettingen.de/pdhC_nucleotide.txt Gene sequence (+200bp) ]''' |
| − | |style="background:#FAF8CC;" align="center"|'''[http://subtiwiki.uni-goettingen.de/ | + | |style="background:#FAF8CC;" align="center"|'''[http://subtiwiki.uni-goettingen.de/pdhC_protein.txt Protein sequence]''' |
|- | |- | ||
| − | |colspan="2" | '''Genetic context''' <br/> [[Image: | + | |colspan="2" | '''Genetic context''' <br/> [[Image:pdhC_context.gif]] |
<div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | <div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | ||
|- | |- | ||
| Line 31: | Line 31: | ||
<br/><br/> | <br/><br/> | ||
| − | |||
=The gene= | =The gene= | ||
| Line 37: | Line 36: | ||
=== Basic information === | === Basic information === | ||
| − | * '''Coordinates:''' | + | * '''Coordinates:''' |
===Phenotypes of a mutant === | ===Phenotypes of a mutant === | ||
| + | |||
| + | defects in sporulation and unable to grow on glucose as single carbon source [http://www.ncbi.nlm.nih.gov/pubmed/11976308 PubMed] | ||
=== Database entries === | === Database entries === | ||
| − | * '''DBTBS entry:''' [http://dbtbs.hgc.jp/COG/prom/ | + | * '''DBTBS entry:''' [http://dbtbs.hgc.jp/COG/prom/pdhABCD.html] |
| − | * '''SubtiList entry:''' [http://genolist.pasteur.fr/SubtiList/genome.cgi?gene_detail+ | + | * '''SubtiList entry:''' [http://genolist.pasteur.fr/SubtiList/genome.cgi?gene_detail+BG10209] |
=== Additional information=== | === Additional information=== | ||
| + | |||
=The protein= | =The protein= | ||
| Line 53: | Line 55: | ||
=== Basic information/ Evolution === | === Basic information/ Evolution === | ||
| − | * '''Catalyzed reaction/ biological activity:''' | + | * '''Catalyzed reaction/ biological activity:''' |
* '''Protein family:''' | * '''Protein family:''' | ||
| Line 64: | Line 66: | ||
* '''Domains:''' | * '''Domains:''' | ||
| − | |||
* '''Modification:''' | * '''Modification:''' | ||
| Line 70: | Line 71: | ||
* '''Cofactor(s):''' | * '''Cofactor(s):''' | ||
| − | * '''Effectors of protein activity:''' | + | * '''Effectors of protein activity:''' |
| − | * '''Interactions:''' | + | * '''Interactions:''' [[PdhA]]-[[PdhB]]-[[PdhC]]-[[PdhD]] |
| − | * '''Localization:''' | + | * '''Localization:''' membrane associated [http://www.ncbi.nlm.nih.gov/pubmed/18763711 PubMed] |
=== Database entries === | === Database entries === | ||
| − | * '''Structure:''' complex with | + | * '''Structure:''' [http://www.rcsb.org/pdb/cgi/explore.cgi?pdbId=1W88 1W88] (E1 in complex with subunit binding domain of E2, ''Geobacillus stearothermophilus''), [http://www.rcsb.org/pdb/cgi/explore.cgi?pdbId=2PDE 2PDE] (peripheral subunit binding domain, ''Geobacillus stearothermophilus''), [http://www.rcsb.org/pdb/cgi/explore.cgi?pdbId=1LAC 1LAC] (lipoyl domain, ''Geobacillus stearothermophilus''), [http://www.rcsb.org/pdb/cgi/explore.cgi?pdbId=1B5S 1B5S] (catalytic domain (residues 184-425) , ''Geobacillus stearothermophilus'') |
| + | * '''Swiss prot entry:''' | ||
| − | * ''' | + | * '''KEGG entry:''' [http://www.genome.jp/dbget-bin/www_bget?bsu+BSU14600] |
| − | * ''' | + | * '''E.C. number:''' 2.3.1.12 |
=== Additional information=== | === Additional information=== | ||
| − | |||
=Expression and regulation= | =Expression and regulation= | ||
| − | * '''Operon:''' | + | * '''Operon:''' ''[[pdhA]]-[[pdhB]]-[[pdhC]]-[[pdhD]]'' |
| − | |||
| − | |||
| − | + | * '''Sigma factor:''' [[SigA]] | |
| − | * ''' | + | * '''Regulation:''' expression activated by glucose (1.94) [http://www.ncbi.nlm.nih.gov/pubmed/12850135 PubMed], weak induction by glucose [http://www.ncbi.nlm.nih.gov/sites/entrez/12850135 PubMed] |
| − | + | * '''Regulatory mechanism:''' | |
| − | |||
| − | * '''Regulatory mechanism:''' | ||
| − | |||
| − | |||
* '''Additional information:''' | * '''Additional information:''' | ||
| Line 107: | Line 102: | ||
=Biological materials = | =Biological materials = | ||
| − | * '''Mutant:''' | + | * '''Mutant:''' |
| − | * '''Expression vector:''' | + | * '''Expression vector:''' |
| − | + | ||
| − | * '''lacZ fusion:''' | + | * '''lacZ fusion:''' |
* '''GFP fusion:''' | * '''GFP fusion:''' | ||
| − | * '''Antibody:''' | + | * '''two-hybrid system:''' |
| + | |||
| + | * '''Antibody:''' | ||
=Labs working on this gene/protein= | =Labs working on this gene/protein= | ||
| − | |||
| − | |||
=Your additional remarks= | =Your additional remarks= | ||
=References= | =References= | ||
| − | + | ||
| − | + | Blencke et al. (2003) Transcriptional profiling of gene expression in response to glucose in ''Bacillus subtilis'': regulation of the central metabolic pathways. ''Metab Eng.'' '''5:''' 133-149 [http://www.ncbi.nlm.nih.gov/pubmed/12850135 PubMed] | |
| − | + | # Hahne et al. (2008) From complementarity to comprehensiveness - targeting the membrane proteome of growing ''Bacillus subtilis'' by divergent approaches. Proteomics 8: 4123-4136 [http://www.ncbi.nlm.nih.gov/pubmed/18763711 PubMed] | |
| − | # | + | # Gao et al. (2002) The E1beta and E2 subunits of the ''Bacillus subtilis'' pyruvate dehydrogenase complex are involved in regulation of sporulation.''J. Bacteriol.'' '''184:''' 2780-2788. [http://www.ncbi.nlm.nih.gov/sites/entrez/PMID PubMed] |
| − | # | + | # Author1, Author2 & Author3 (year) Title ''Journal'' '''volume:''' page-page. [http://www.ncbi.nlm.nih.gov/sites/entrez/PMID PubMed] |
| − | |||
| − | |||
| − | # | ||
| − | |||
| − | |||
| − | |||
Revision as of 23:24, 1 April 2009
- Description: pyruvate dehydrogenase (dihydrolipoamide acetyltransferase E2 subunit)
| Gene name | pdhC |
| Synonyms | |
| Essential | no |
| Product | pyruvate dehydrogenase (dihydrolipoamide acetyltransferase E2 subunit) |
| Function | links glycolysis and TCA cycle |
| MW, pI | 47 kDa, 4.855 |
| Gene length, protein length | 1326 bp, 442 aa |
| Immediate neighbours | pdhB, pdhD |
| Gene sequence (+200bp) | Protein sequence |
Genetic context 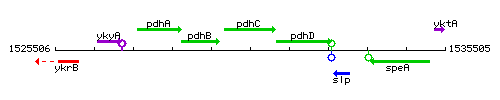
This image was kindly provided by SubtiList
| |
Contents
The gene
Basic information
- Coordinates:
Phenotypes of a mutant
defects in sporulation and unable to grow on glucose as single carbon source PubMed
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family:
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Localization: membrane associated PubMed
Database entries
- Structure: 1W88 (E1 in complex with subunit binding domain of E2, Geobacillus stearothermophilus), 2PDE (peripheral subunit binding domain, Geobacillus stearothermophilus), 1LAC (lipoyl domain, Geobacillus stearothermophilus), 1B5S (catalytic domain (residues 184-425) , Geobacillus stearothermophilus)
- Swiss prot entry:
- KEGG entry: [3]
- E.C. number: 2.3.1.12
Additional information
Expression and regulation
- Sigma factor: SigA
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Blencke et al. (2003) Transcriptional profiling of gene expression in response to glucose in Bacillus subtilis: regulation of the central metabolic pathways. Metab Eng. 5: 133-149 PubMed
- Hahne et al. (2008) From complementarity to comprehensiveness - targeting the membrane proteome of growing Bacillus subtilis by divergent approaches. Proteomics 8: 4123-4136 PubMed
- Gao et al. (2002) The E1beta and E2 subunits of the Bacillus subtilis pyruvate dehydrogenase complex are involved in regulation of sporulation.J. Bacteriol. 184: 2780-2788. PubMed
- Author1, Author2 & Author3 (year) Title Journal volume: page-page. PubMed