Difference between revisions of "ClpX"
| Line 56: | Line 56: | ||
===Phenotypes of a mutant === | ===Phenotypes of a mutant === | ||
| + | * increased thermotolerance due to increased stabiliy of [[Spx]] and thus increased expression of ''[[trxA]]'' {{PubMed|24417481}} | ||
=== Database entries === | === Database entries === | ||
| Line 64: | Line 65: | ||
=== Additional information=== | === Additional information=== | ||
| − | |||
| − | |||
| − | |||
=The protein= | =The protein= | ||
| Line 85: | Line 83: | ||
* '''Kinetic information:''' | * '''Kinetic information:''' | ||
| − | * '''Domains:''' AAA-ATPase [http://pfam.sanger.ac.uk/family?acc=PF07724 PFAM] | + | * '''[[Domains]]:''' |
| + | ** AAA-ATPase [http://pfam.sanger.ac.uk/family?acc=PF07724 PFAM] | ||
| + | ** Zinc finger [http://pfam.sanger.ac.uk/family?acc=PF06689 PFAM] | ||
* '''Modification:''' | * '''Modification:''' | ||
| − | * ''' | + | * '''[[Cofactors]]:''' |
* '''Effectors of protein activity:''' | * '''Effectors of protein activity:''' | ||
| Line 150: | Line 150: | ||
=References= | =References= | ||
==Reviews== | ==Reviews== | ||
| − | + | <pubmed>23375660 19680248 17302811 23479438,19609260</pubmed> | |
| − | <pubmed>23375660 19680248 17302811</pubmed> | ||
==Original Publications== | ==Original Publications== | ||
| − | + | <pubmed>12761164 10809708,9643546,11807061,14679237,18689476,16899079,8973311, 19136590 , 11325926 8973311 9852015 18689473 20525796 15948963 18786145 24417481</pubmed> | |
| − | <pubmed>12761164 10809708,9643546,11807061,14679237,18689476,16899079,8973311, 19136590 , 11325926 8973311 9852015 18689473 20525796 15948963 </pubmed> | ||
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 10:32, 16 January 2014
- Description: ATP-dependent Clp protease ATP-binding subunit (class III heat-shock protein)
| Gene name | clpX |
| Synonyms | |
| Essential | no |
| Product | ATP-dependent Clp protease ATP-binding subunit |
| Function | protein degradation |
| Gene expression levels in SubtiExpress: clpX | |
| Interactions involving this protein in SubtInteract: ClpX | |
| Metabolic function and regulation of this protein in SubtiPathways: clpX | |
| MW, pI | 46 kDa, 4.645 |
| Gene length, protein length | 1260 bp, 420 aa |
| Immediate neighbours | lonB, tig |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 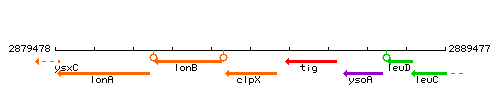
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed
| |
Contents
Categories containing this gene/protein
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU28220
Phenotypes of a mutant
- increased thermotolerance due to increased stabiliy of Spx and thus increased expression of trxA PubMed
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: ATPase/chaperone
- Protein family: clpX chaperone family (according to Swiss-Prot) ClpX (IP004487) InterPro, AAA+ -type ATPase (IPR013093) InterPro (PF07724) PFAM
Targets of ClpX-ClpP-dependent protein degradation
Extended information on the protein
- Kinetic information:
- Modification:
- Effectors of protein activity:
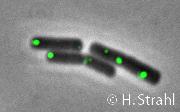
- Localization: cytoplasmic polar clusters, excluded from the nucleoid, induced clustering upon heat shock, colocalization with ClpP PubMed
Database entries
- Structure: homologue structure resolved 1UM8, structural model of B. subtilis ClpX available from hstrahl
- UniProt: P50866
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Operon: clpX PubMed
- Additional information:
- The mRNA has a long 5' leader region. This may indicate RNA-based regulation PubMed
Biological materials
- Mutant: clpX::kan, clpX::spec and clpX::cat available from the Hamoen] Lab
- Expression vector:
- lacZ fusion:
- GFP fusion: C-terminal GFP fusions (both single copy and 2th copy in amyE locus, also as CFP and YFP variants) available from the Hamoen] Lab
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Leendert Hamoen, Newcastle University, UK homepage
Your additional remarks
References
Reviews
Original Publications