Difference between revisions of "SinI"
Czschiedrich (talk | contribs) (→Biological materials) |
|||
| Line 139: | Line 139: | ||
** DS91 (spc) NCIB3610 derivate, available in [[Stülke]] lab | ** DS91 (spc) NCIB3610 derivate, available in [[Stülke]] lab | ||
** 1S98 ( ''sinI''::''kan''), {{PubMed|11466285}}, available at [http://pasture.asc.ohio-state.edu/BGSC/getdetail.cfm?bgscid=1S98&Search=1S98 BGSC] | ** 1S98 ( ''sinI''::''kan''), {{PubMed|11466285}}, available at [http://pasture.asc.ohio-state.edu/BGSC/getdetail.cfm?bgscid=1S98&Search=1S98 BGSC] | ||
| + | ** GP1663 (''yghG-sinI-sinR-tasA''), available in [[Stülke]] lab | ||
* '''Expression vector:''' | * '''Expression vector:''' | ||
Revision as of 14:26, 1 November 2013
| Gene name | sinI |
| Synonyms | |
| Essential | no |
| Product | antagonist of SinR |
| Function | control of biofilm formation |
| Gene expression levels in SubtiExpress: sinI | |
| Interactions involving this protein in SubtInteract: SinI | |
| Regulation of this protein in SubtiPathways: Biofilm | |
| MW, pI | 6 kDa, 6.333 |
| Gene length, protein length | 171 bp, 57 aa |
| Immediate neighbours | yqhG, sinR |
| Sequences | Protein DNA DNA_with_flanks |
Genetic context 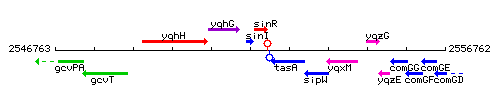
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed
| |
Contents
Categories containing this gene/protein
transcription factors and their control, transition state regulators, biofilm formation
This gene is a member of the following regulons
AbrB regulon, ScoC regulon, Spo0A regulon
The gene
Basic information
- Locus tag: BSU24600
Phenotypes of a mutant
- altered cell death pattern in colonies PubMed
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family:
- Paralogous protein(s): SlrA
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
Database entries
- UniProt: P23308
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Regulation:
- Regulatory mechanism:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system: B. pertussis adenylate cyclase-based bacterial two hybrid system (BACTH), available in Stülke lab
- FLAG-tag construct (C-term): GP935 (kan), available in Stülke lab
- Antibody:
Labs working on this gene/ protein
Your additional remarks
References
Reviews
Modelling of the SinI/SinR switch
Original publications