|
|
| Line 1: |
Line 1: |
| − | * '''Description:''' HPr, General component of the sugar phosphotransferase system (PTS). <br/><br/> | + | * '''Description:''' dihydrolipoamide dehydrogenase E3 subunit of both pyruvate dehydrogenase and 2-oxoglutarate dehydrogenase complexes <br/><br/> |
| | | | |
| | {| align="right" border="1" cellpadding="2" | | {| align="right" border="1" cellpadding="2" |
| | |- | | |- |
| | |style="background:#ABCDEF;" align="center"|'''Gene name''' | | |style="background:#ABCDEF;" align="center"|'''Gene name''' |
| − | |''ptsH'' | + | |''pdhD'' |
| | |- | | |- |
| − | |style="background:#ABCDEF;" align="center"| '''Synonyms''' || '' '' | + | |style="background:#ABCDEF;" align="center"| '''Synonyms''' || ''citL '' |
| | |- | | |- |
| | |style="background:#ABCDEF;" align="center"| '''Essential''' || no | | |style="background:#ABCDEF;" align="center"| '''Essential''' || no |
| | |- | | |- |
| − | |style="background:#ABCDEF;" align="center"| '''Product''' || histidine-containing phosphocarrier protein HPr of the PTS | + | |style="background:#ABCDEF;" align="center"| '''Product''' || dihydrolipoamide dehydrogenase E3 subunit<br/> of both pyruvate dehydrogenase and 2-oxoglutarate<br/> dehydrogenase complexes |
| | |- | | |- |
| − | |style="background:#ABCDEF;" align="center"|'''Function''' || PTS-dependent sugar trnasport and carbon catabolite repression | + | |style="background:#ABCDEF;" align="center"|'''Function''' || links glycolysis and TCA cycle, enzyme in TCA cycle |
| | |- | | |- |
| − | |style="background:#ABCDEF;" align="center"| '''MW, pI''' || 9,1 kDa, 4.58 | + | |style="background:#ABCDEF;" align="center"| '''MW, pI''' || 49 kDa, 4.76 |
| | |- | | |- |
| − | |style="background:#ABCDEF;" align="center"| '''Gene length, protein length''' || 264 bp, 88 amino acids | + | |style="background:#ABCDEF;" align="center"| '''Gene length, protein length''' || 1410 bp, 470 aa |
| | |- | | |- |
| − | |style="background:#ABCDEF;" align="center"|'''Immediate neighbours''' || ''[[ptsG]]'', ''[[ptsI]]'' | + | |style="background:#ABCDEF;" align="center"|'''Immediate neighbours''' || ''[[pdhC]]'', ''[[slp]]'' |
| | |- | | |- |
| − | |style="background:#FAF8CC;" align="center"|'''[http://subtiwiki.uni-goettingen.de/ptsH_nucleotide.txt Gene sequence (+200bp) ]''' | + | |style="background:#FAF8CC;" align="center"|'''[http://subtiwiki.uni-goettingen.de/pdhD_nucleotide.txt Gene sequence (+200bp) ]''' |
| − | |style="background:#FAF8CC;" align="center"|'''[http://subtiwiki.uni-goettingen.de/ptsH_protein.txt Protein sequence]''' | + | |style="background:#FAF8CC;" align="center"|'''[http://subtiwiki.uni-goettingen.de/pdhD_protein.txt Protein sequence]''' |
| | |- | | |- |
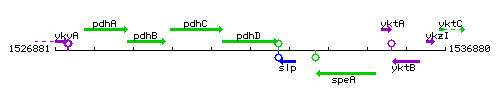
| − | |colspan="2" | '''Genetic context''' <br/> [[Image:ptsH_context.gif]] | + | |colspan="2" | '''Genetic context''' <br/> [[Image:pdhD_context.gif]] |
| | <div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> | | <div align="right"> <small>This image was kindly provided by [http://genolist.pasteur.fr/SubtiList/ SubtiList]</small></div> |
| | |- | | |- |
| Line 30: |
Line 30: |
| | __TOC__ | | __TOC__ |
| | | | |
| − | <br/><br/> | + | <br/><br/><br/><br/> |
| − | | |
| | | | |
| | =The gene= | | =The gene= |
| Line 37: |
Line 36: |
| | === Basic information === | | === Basic information === |
| | | | |
| − | * '''Coordinates:''' 1458693 - 1458956 | + | * '''Coordinates:''' |
| | | | |
| | ===Phenotypes of a mutant === | | ===Phenotypes of a mutant === |
| | + | |
| | + | defects in sporulation and unable to grow on glucose as single carbon source [http://www.ncbi.nlm.nih.gov/pubmed/11976308 PubMed] |
| | | | |
| | === Database entries === | | === Database entries === |
| | | | |
| − | * '''DBTBS entry:''' [http://dbtbs.hgc.jp/COG/prom/ptsGHI.html] | + | * '''DBTBS entry:''' [http://dbtbs.hgc.jp/COG/prom/pdhABCD.html] |
| | | | |
| − | * '''SubtiList entry:'''[http://genolist.pasteur.fr/SubtiList/genome.cgi?gene_detail+BG10200] | + | * '''SubtiList entry:''' [http://genolist.pasteur.fr/SubtiList/genome.cgi?gene_detail+BG10210] |
| | | | |
| | === Additional information=== | | === Additional information=== |
| | + | |
| | | | |
| | =The protein= | | =The protein= |
| Line 53: |
Line 55: |
| | === Basic information/ Evolution === | | === Basic information/ Evolution === |
| | | | |
| − | * '''Catalyzed reaction/ biological activity:''' Protein HPr N(pi)-phospho-L-histidine + protein EIIA = protein [[PtsH|HPr]] + protein EIIA N(tau)-phospho-L-histidine | + | * '''Catalyzed reaction/ biological activity:''' |
| | | | |
| − | * '''Protein family:''' HPr family | + | * '''Protein family:''' |
| | | | |
| − | * '''Paralogous protein(s):''' [[Crh]] | + | * '''Paralogous protein(s):''' |
| | | | |
| | === Extended information on the protein === | | === Extended information on the protein === |
| Line 63: |
Line 65: |
| | * '''Kinetic information:''' | | * '''Kinetic information:''' |
| | | | |
| − | * '''Domains:''' HPr Domain (2–88) | + | * '''Domains:''' |
| | | | |
| − | * '''Modification:''' phosphorylations: transient phosphorylation by [[PtsI |Enzyme I]] of the PTS on His-15, regulatory phosphorylation on Ser-46 by [[HprK]] [http://www.ncbi.nlm.nih.gov/sites/entrez/2507315 PubMed], weak phosphorylation on Ser-12 [http://www.ncbi.nlm.nih.gov/sites/entrez/17218307 PubMed], an extensive study on ''in vivo'' HPr phosphorylation can be found in Singh et al. (2008) [http://www.ncbi.nlm.nih.gov/sites/entrez/18757537 PubMed] | + | * '''Modification:''' |
| | | | |
| | * '''Cofactor(s):''' | | * '''Cofactor(s):''' |
| Line 71: |
Line 73: |
| | * '''Effectors of protein activity:''' | | * '''Effectors of protein activity:''' |
| | | | |
| − | * '''Interactions:''' [[GapA]]-[[PtsH|HPr]] [http://www.ncbi.nlm.nih.gov/sites/entrez/17142398 PubMed], [[PtsH|HPr]]-[[MtlR]], [[PtsH|HPr]]-[[LicR]], [[PtsH|HPr]]-[[LevR]],[[PtsH|HPr]]-[[ManR]], [[YesS]]-[[PtsH|HPr]] (HPr-His-P), [[PtsH|HPr]]-[[CcpA]] [http://www.ncbi.nlm.nih.gov/sites/entrez/12432959 PubMed], [[PtsH|HPr]]-[[RbsR]] [http://www.ncbi.nlm.nih.gov/sites/entrez/16519689 PubMed], [[HprK]]-[[PtsH|HPr]] [http://www.ncbi.nlm.nih.gov/sites/entrez/12009882 PubMed] | + | * '''Interactions:''' [[PdhA]]-[[PdhB]]-[[PdhC]]-[[PdhD]], [[OdhA]]-[[OdhB]]-[[PdhD]] |
| | | | |
| − | * '''Localization:''' Cytoplasm [http://www.ncbi.nlm.nih.gov/sites/entrez/16395550 PubMed] | + | * '''Localization:''' |
| | | | |
| | === Database entries === | | === Database entries === |
| | | | |
| − | * '''Structure:''' [http://www.ncbi.nlm.nih.gov/Structure/mmdb/mmdbsrv.cgi?Dopt=s&uid=58038 NCBI], complex of L. Casei HprK with B. Subtilis HPr [http://www.ncbi.nlm.nih.gov/Structure/mmdb/mmdbsrv.cgi?Dopt=s&uid=20417 NCBI], complex of L. Casei HprK with B. Subtilis HPr-Ser-P [http://www.ncbi.nlm.nih.gov/Structure/mmdb/mmdbsrv.cgi?Dopt=s&uid=20418 NCBI] | + | * '''Structure:''' [http://www.rcsb.org/pdb/cgi/explore.cgi?pdbId=1EBD 1EBD] (complex with binding domain of dihydrolipoamide acetylase, ''Geobacillus stearothermophilus'') |
| | | | |
| − | * '''Swiss prot entry:''' [http://www.expasy.ch/cgi-bin/sprot-search-ac?P08877] | + | * '''Swiss prot entry:''' |
| | | | |
| − | * '''KEGG entry:''' [http://www.genome.jp/dbget-bin/www_bget?bsu+BSU13900] | + | * '''KEGG entry:''' [http://www.genome.jp/dbget-bin/www_bget?bsu+BSU14610] |
| | | | |
| − | * '''E.C. number:''' [http://www.expasy.org/enzyme/2.7.11.-] | + | * '''E.C. number:''' 1.8.1.4 |
| | | | |
| | === Additional information=== | | === Additional information=== |
| − |
| |
| | | | |
| | =Expression and regulation= | | =Expression and regulation= |
| | | | |
| − | * '''Operon:''' | + | * '''Operon:''' ''[[pdhA]]-[[pdhB]]-[[pdhC]]-[[pdhD]]'' |
| − | **''[[ptsG]]-[[ptsH]]-[[ptsI]]''
| |
| − | **''ptsH-[[ptsI]]''
| |
| | | | |
| − | * '''Sigma factor:''' [[SigA]] [http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Abstract&list_uids=+11902727 PubMed] | + | * '''Sigma factor:''' [[SigA]] |
| | | | |
| − | * '''Regulation:''' expression activated by glucose (1.98) [http://www.ncbi.nlm.nih.gov/pubmed/12850135 PubMed] induction by glucose (''[[ptsG]]''), constitutive (''[[ptsH]]'') | + | * '''Regulation:''' expression activated by glucose (2.03) [http://www.ncbi.nlm.nih.gov/pubmed/12850135 PubMed] weak induction by glucose [http://www.ncbi.nlm.nih.gov/sites/entrez/12850135 PubMed] |
| | | | |
| − | * '''Regulatory mechanism:''' ''[[ptsG]]'': transcriptional antitermination via the [[GlcT]]-dependent RNA-switch | + | * '''Regulatory mechanism:''' |
| | | | |
| | * '''Additional information:''' | | * '''Additional information:''' |
| Line 104: |
Line 103: |
| | =Biological materials = | | =Biological materials = |
| | | | |
| − | * '''Mutant:''' MZ303 (cat), GP507 ptsH1 (S46A), GP506 (ptsH-H15A), available in [[Stülke]] lab | + | * '''Mutant:''' |
| | | | |
| − | * '''Expression vector:''' pGP438 (with N-terminal Strep-tag, in [[pGP172]]), pAG2 (His-tag) pGP371(ptsH-S46A, with His-tag, in [[pWH844]]), available in [[Stülke]] | + | * '''Expression vector:''' |
| − |
| + | |
| | * '''lacZ fusion:''' | | * '''lacZ fusion:''' |
| | | | |
| | * '''GFP fusion:''' | | * '''GFP fusion:''' |
| | | | |
| − | * '''two-hybrid system:''' B. pertussis adenylate cyclase-based bacterial two hybrid system ([[BACTH]]), available in [[Boris Görke| Görke]] lab | + | * '''two-hybrid system:''' |
| | | | |
| − | * '''Antibody:''' available in [[Stülke]] lab | + | * '''Antibody:''' |
| | | | |
| | =Labs working on this gene/protein= | | =Labs working on this gene/protein= |
| − |
| |
| − | [[Josef Deutscher]], Paris-Grignon, France
| |
| − |
| |
| − | [[Stülke|Jörg Stülke]], University of Göttingen, Germany [http://wwwuser.gwdg.de/~genmibio/stuelke.html Homepage]
| |
| − |
| |
| − | [[Wolfgang Hillen]], Erlangen University, Germany [http://www.biologie.uni-erlangen.de/mibi/index2.html Homepage]
| |
| − |
| |
| − | [[Richard Brennan]], Houston, Texas, USA [http://www.mdanderson.org/departments/biochem/display.cfm?id=556ef368-6c81-4043-b74f350d41dd06cb&method=displayfull&pn=a8427ebd-d0ff-11d4-80fd00508b603a14 Homepage]
| |
| − |
| |
| − | [[Boris Görke]], University of Göttingen, Germany [http://wwwuser.gwdg.de/~genmibio/goerke.html Homepage]
| |
| − |
| |
| − | [[Anne Galinier]], University of Marseille, France
| |
| | | | |
| | =Your additional remarks= | | =Your additional remarks= |
| Line 134: |
Line 121: |
| | =References= | | =References= |
| | | | |
| − | # Macek B, Mijakovic I, Olsen JV (2007) The serine/threonine/tyrosine phosphoproteome of the model bacterium Bacillus subtilis. ''Mol Cell Proteomics'' '''6(4):''' 697-707. [http://www.ncbi.nlm.nih.gov/sites/entrez/17218307 PubMed] | + | # Gao et al. (2002) The E1beta and E2 subunits of the ''Bacillus subtilis'' pyruvate dehydrogenase complex are involved in regulation of sporulation.''J. Bacteriol.'' '''184:''' 2780-2788. [http://www.ncbi.nlm.nih.gov/sites/entrez/PMID PubMed] |
| − | # Müller W, Horstmann N, Hillen W (2006) The transcription regulator RbsR represents a novel interaction partner of the phosphoprotein HPr-Ser46-P in Bacillus subtilis ''FEBS J.'' '''273(6):''' 1251-61. [http://www.ncbi.nlm.nih.gov/sites/entrez/16519689 PubMed]
| + | # Author1, Author2 & Author3 (year) Title ''Journal'' '''volume:''' page-page. [http://www.ncbi.nlm.nih.gov/sites/entrez/PMID PubMed] |
| − | # Pompeo ''et al.'' (2007) Interaction of GapA with HPr and its homologue, Crh: Novel levels of regulation of a key step of glycolysis in ''Bacillus subtilis''? J Bacteriol 189, 1154-1157.[http://www.ncbi.nlm.nih.gov/sites/entrez/17142398 PubMed]
| |
| − | # Fieulaine, S., Morera, S., Poncet, S., Mijakovic, I., Galinier, A., Janin, J., Deutscher, J., and Nessler, S. (2002) X-ray structure of a bifunctional protein kinase in complex with its protein substrate HPr. Proc Natl Acad Sci U S A 99: 13437-13441. [http://www.ncbi.nlm.nih.gov/sites/entrez/12359875 PubMed]
| |
| − | # Arnaud M, Vary P, Zagorec M, Klier A, Débarbouillé M, Postma P, Rapoport G (1992) Regulation of the sacPA operon of Bacillus subtilis: identification of phosphotransferase system components involved in SacT activity. J Bacteriol 174:3161-3170. [http://www.ncbi.nlm.nih.gov/sites/entrez/1577686 PubMed]
| |
| − | # Deutscher, J., Kessler, U., Alpert, C. A., and Hengstenberg, W. (1984) Bacterial phosphoenolpyruvate-dependent phosphotransferase system: P-ser-HPr and its possible regulatory function. Biochemistry 23: 4455-4460. [http://pubs.acs.org/doi/abs/10.1021/bi00314a033 DOI:10.1021/bi00314a033]
| |
| − | # Deutscher, J., Küster, E., Bergstedt, U., Charrier, V., and Hillen, W. (1995) Protein kinase-dependent HPr/CcpA interaction links glycolytic activity to carbon catabolite repression in Gram-positive bacteria. Mol. Microbiol. 15: 1049-1053. [http://www.ncbi.nlm.nih.gov/sites/entrez/7623661 PubMed]
| |
| − | # Eisermann, R., Deutscher, J., Gonzy-Tréboul, G., and Hengstenberg, W. (1988) Site-directed mutagenesis with the ptsH gene of Bacillus subtilis. J Biol Chem 263: 17050-17054. [http://www.ncbi.nlm.nih.gov/sites/entrez/2846556 PubMed]
| |
| − | # Frisby, D., and Zuber, P. 1994. Mutations in pts cause catabolite-resistant sporulation and altered regulation of spo0H in Bacillus subtilis. J. Bacteriol. 176: 2587-2595. [http://www.ncbi.nlm.nih.gov/sites/entrez/8169206 PubMed]
| |
| − | # Galinier A, Deutscher J, Martin-Verstraete I: (1999) Phosphorylation of either Crh or HPr mediates binding of CcpA to the Bacillus subtilis xyn cre and catabolite repression of the xyn operon. J Mol Biol , 286:307-314. [http://www.ncbi.nlm.nih.gov/sites/entrez/9973552 PubMed]
| |
| − | # Görke, B., Fraysse, L. & Galinier, A. (2004) Drastic differences in Crh and HPr synthesis levels reflect their different impacts on catabolite repression in Bacillus subtilis. J. Bacteriol. 186, 2992-2995 . [http://www.ncbi.nlm.nih.gov/sites/entrez/15126459 PubMed]
| |
| − | # Lindner, C., Galinier, A., Hecker, M. & Deutscher, J. (1999) Regulation of the activity of the Bacillus subtilis antiterminator LicT by multiple PEP-dependent, enzyme I- and HPr-catalysed phosphorylation. Mol. Microbiol. 31, 995-1006 . [http://www.ncbi.nlm.nih.gov/sites/entrez/10048041 PubMed]
| |
| − | # Lindner, C., Hecker, M., Le Coq, D. & Deutscher, J. (2002) Bacillus subtilis mutant LicT antiterminators exhibiting enzyme I- and HPr-independent antitermination affect catabolite repression of the bglPH operon. J. Bacteriol. 184, 4819-4828 . [http://www.ncbi.nlm.nih.gov/sites/entrez/12169607 PubMed]
| |
| − | # Martin-Verstraete, I., Charrier, V., Stülke, J., Galinier, A., Erni, B., Rapoport, G., & Deutscher, J. (1998) Antagonistic effects of dual PTS catalyzed phosphorylation on the Bacillus subtilis transcriptional activator LevR. Mol. Microbiol. 28: 293-303. [http://www.ncbi.nlm.nih.gov/sites/entrez/9622354 PubMed]
| |
| − | # Martin-Verstraete, I., Deutscher, J., and Galinier, A. (1999) Phosphorylation of HPr and Crh by HprK, early steps in the catabolite repression signalling pathway for the Bacillus subtilis levanase operon. J Bacteriol 181: 2966-2969. [http://www.ncbi.nlm.nih.gov/sites/entrez/10217795 PubMed]
| |
| − | # Reizer, J., Sutrina, S. L., Saier, Jr., M. H., Stewart, G. C., Peterkofsky, A., and Reddy, P. (1989) Mechanistic and physiological consequences of HPr(Ser) phosphorylation on the activities of the phosphoenolpyruvate:sugar phosphotransferase system in Gram-positive bacteria: studies with site-specific mutants of HPr. EMBO J 8: 2111-2120. [http://www.ncbi.nlm.nih.gov/sites/entrez/2507315 PubMed]
| |
| − | # Schmalisch, M., Bachem, S. & Stülke, J. (2003) Control of the Bacillus subtilis antiterminator protein GlcT by phosphorylation: Elucidation of the phosphorylation chain leading to inactivation of GlcT. J. Biol. Chem. 278: 51108-51115. [http://www.ncbi.nlm.nih.gov/sites/entrez/14527945 PubMed]
| |
| − | # Schumacher, M. A. et al. (2004) Structural basis for allosteric control of the transcription regulator CcpA by the phosphoprotein HPr-Ser46-P. Cell 118, 731-741 . [http://www.ncbi.nlm.nih.gov/sites/entrez/15369672 PubMed]
| |
| − | # Singh, K. D., Halbedel, S., Görke, B. & Stülke, J. (2007) Control of the phosphorylation state of the HPr protein of the phosphotransferase system in Bacillus subtilis: implication of the protein phosphatase PrpC. J. Mol. Microbiol. Biotechnol. 13: 165-171. [http://www.ncbi.nlm.nih.gov/sites/entrez/17693724 PubMed] | |
| − | # Singh, K. D., Schmalisch, M. H., Stülke, J. & Görke, B. (2008) Carbon catabolite repression in Bacillus subtilis: A quantitative analysis of repression exerted by different carbon sources. J. Bacteriol. 190: 7275-7284. [http://www.ncbi.nlm.nih.gov/sites/entrez/18757537 PubMed]
| |
| − | # Stülke, J., Martin-Verstraete, I., Charrier, V., Klier, A., Deutscher, J. & Rapoport, G. (1995) The HPr protein of the phosphotransferase system links induction and catabolite repression of the Bacillus subtilis levanase operon. J. Bacteriol. 177: 6928-6936. [http://www.ncbi.nlm.nih.gov/sites/entrez/7592487 PubMed]
| |
| − | # Tortosa, P., Aymerich, S., Lindner, C., Saier, M.H., Jr., Reizer, J. and Le Coq, D. (1997) Multiple phosphorylation of SacY, a Bacillus subtilis antiterminator negatively controlled by the phosphotransferase system. J. Biol. Chem. 272, 17230-17237. [http://www.ncbi.nlm.nih.gov/sites/entrez/9202047 PubMed]
| |
| − | # Charrier V, Buckley E, Parsonage D, Galinier A, Darbon E, Jaquinod M, Forest E, Deutscher J, Claiborne A (1997) Cloning and sequencing of two enterococcal ''glpK'' genes and regulation of the encoded glycerol kinases by phosphoenolpyruvate-dependent, phosphotransferase system-catalyzed phosphorylation of a single histidyl residue. J Biol Chem 272:14166-14174. [http://www.ncbi.nlm.nih.gov/sites/entrez/9162046 PubMed]
| |
| − | # Darbon E, Servant P, Poncet S, Deutscher J (2002) Antitermination by GlpP, catabolite repression via CcpA and inducer exclusion triggered by P~GlpK dephosphorylation control ''Bacillus subtilis glpFK'' expression. Mol Microbiol 43:1039-1052. [http://www.ncbi.nlm.nih.gov/sites/entrez/11929549 PubMed]
| |
| − | # Jones, B.E., Rajagopal, P., and Klevit, R.E. (1997) Phosphorylation on histidine is accompanied by localized structural changes in the phosphocarrier protein, HPr from Bacillus subtilis. Protein Sci 6: 2107-2119. [http://www.ncbi.nlm.nih.gov/sites/entrez/9336834 PubMed]
| |
| − | # Rajagopal, P., Waygood, E.B., and Klevit, R.E. (1994) Structural consequences of histidine phosphorylation: NMR characterization of the phosphohistidine form of histidine-containing protein from Bacillus subtilis and Escherichia coli. Biochemistry 33: 15271-15282. [http://www.ncbi.nlm.nih.gov/sites/entrez/7803390 PubMed]
| |