Difference between revisions of "Bmr"
| Line 84: | Line 84: | ||
* '''Effectors of protein activity:''' | * '''Effectors of protein activity:''' | ||
| − | * '''Interactions:''' | + | * '''[[SubtInteract|Interactions]]:''' |
| − | * '''Localization:''' | + | * '''[[Localization]]:''' |
=== Database entries === | === Database entries === | ||
Revision as of 10:24, 16 July 2011
- Description: general stress protein, multidrug-efflux transporter
| Gene name | bmr |
| Synonyms | bmr1 |
| Essential | no |
| Product | multidrug-efflux transporter |
| Function | multidrug resistance |
| MW, pI | 42 kDa, 9.696 |
| Gene length, protein length | 1167 bp, 389 aa |
| Immediate neighbours | yqiW, bmrR |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 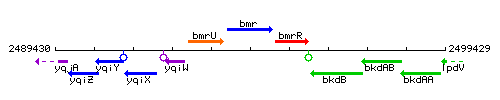
This image was kindly provided by SubtiList
| |
Contents
Categories containing this gene/protein
transporters/ other, general stress proteins (controlled by SigB), resistance against toxins/ antibiotics
This gene is a member of the following regulons
BmrR regulon, Mta regulon, SigB regulon
The gene
Basic information
- Locus tag: BSU24010
Phenotypes of a mutant
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family: TCR/tet family (according to Swiss-Prot)
- Paralogous protein(s): Blt
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
Database entries
- Structure:
- UniProt: P33449
- KEGG entry: [3]
- E.C. number:
Additional information
Expression and regulation
- Regulation:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References
Muthiah Kumaraswami, Kate J Newberry, Richard G Brennan
Conformational plasticity of the coiled-coil domain of BmrR is required for bmr operator binding: the structure of unliganded BmrR.
J Mol Biol: 2010, 398(2);264-75
[PubMed:20230832]
[WorldCat.org]
[DOI]
(I p)
A Petersohn, M Brigulla, S Haas, J D Hoheisel, U Völker, M Hecker
Global analysis of the general stress response of Bacillus subtilis.
J Bacteriol: 2001, 183(19);5617-31
[PubMed:11544224]
[WorldCat.org]
[DOI]
(P p)
Anja Petersohn, Haike Antelmann, Ulf Gerth, Michael Hecker
Identification and transcriptional analysis of new members of the sigmaB regulon in Bacillus subtilis.
Microbiology (Reading): 1999, 145 ( Pt 4);869-880
[PubMed:10220166]
[WorldCat.org]
[DOI]
(P p)
N N Baranova, A Danchin, A A Neyfakh
Mta, a global MerR-type regulator of the Bacillus subtilis multidrug-efflux transporters.
Mol Microbiol: 1999, 31(5);1549-59
[PubMed:10200972]
[WorldCat.org]
[DOI]
(P p)
M Ahmed, L Lyass, P N Markham, S S Taylor, N Vázquez-Laslop, A A Neyfakh
Two highly similar multidrug transporters of Bacillus subtilis whose expression is differentially regulated.
J Bacteriol: 1995, 177(14);3904-10
[PubMed:7608059]
[WorldCat.org]
[DOI]
(P p)
M Ahmed, C M Borsch, S S Taylor, N Vázquez-Laslop, A A Neyfakh
A protein that activates expression of a multidrug efflux transporter upon binding the transporter substrates.
J Biol Chem: 1994, 269(45);28506-13
[PubMed:7961792]
[WorldCat.org]
(P p)