CcpA
- Description: Carbon catabolite control protein A, involved in glucose regulation of many genes; represses catabolic genes and activates genes involved in excretion of excess carbon
| Gene name | ccpA |
| Synonyms | graR, alsA, amyR |
| Essential | no |
| Product | transcriptional regulator (LacI family) |
| Function | mediates carbon catabolite repression (CCR) |
| Gene expression levels in SubtiExpress: ccpA | |
| Interactions involving this protein in SubtInteract: CcpA | |
| Metabolic function and regulation of this protein in SubtiPathways: Nucleoside catabolism, Nucleotides (regulation), Ile, Leu, Val, His, Coenzyme A, Central C-metabolism | |
| MW, pI | 36,8 kDa, 5.06 |
| Gene length, protein length | 1002 bp, 334 amino acids |
| Immediate neighbours | motP, aroA |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 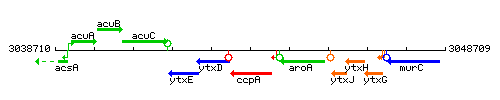
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed
| |
Contents
- 1 Categories containing this gene/protein
- 2 This gene is a member of the following regulons
- 3 The CcpA regulon
- 4 The gene
- 5 The protein
- 6 Expression and regulation
- 7 Biological materials
- 8 Labs working on this gene/protein
- 9 Your additional remarks
- 10 References
- 10.1 Reviews
- 10.2 General and physiological studies
- 10.3 Global analyses (proteome, transcriptome, ChIP-chip)
- 10.4 Repression of target genes by CcpA
- 10.5 Positive regulation of gene expression by CcpA
- 10.6 Control of CcpA activity
- 10.7 CcpA-DNA interaction
- 10.8 Functional analysis of CcpA
- 10.9 Structural analyses
Categories containing this gene/protein
- see also: glutamate metabolism
This gene is a member of the following regulons
The CcpA regulon
The gene
Basic information
- Locus tag: BSU29740
Phenotypes of a mutant
Loss of carbon catabolite repression. Loss of PTS-dependent sugar transport due to excessive phosphorylation of HPr by HprK. The mutant is unable to grow on a minimal medium with glucose and ammonium as the only sources of carbon and nitrogen, respectively.
Database entries
- DBTBS entry: [1]
- SubtiList entry: [2]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: transcriptional regulator of carbon catabolite repression (CCR)
- Protein family: LacI family
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- HTH LacI-type Domain (1 – 58)
- DNA binding Domain (6 – 25)
- Modification:
- Effectors of protein activity:glucose-6-phosphate, fructose-1,6-bisphosphate Pubmed
Database entries
- Structure:
- 2JCG (Apoprotein from Bacillus megaterium)
- CcpA-Crh-DNA-complex NCBI
- complex with P-Ser-HPr and sulphate ions NCBI
- 3OQM (complex of B. subtilis CcpA with P-Ser-HPr and the ackA operator site)
- 3OQN (complex of B. subtilis CcpA with P-Ser-HPr and the gntR operator site)
- 3OQO (complex of B. subtilis CcpA with P-Ser-HPr and a optimal synthetic operator site)
- UniProt: P25144
- KEGG entry: [3]
Additional information
Expression and regulation
- Sigma factor:
- Regulation: constitutively expressed PubMed
- Additional information: there are about 3.000 molecules of CcpA per cell PubMed, this corresponds to a concentration of 3 myM (according to PubMed)
Biological materials
- Mutant: QB5407 (spc), GP302 (erm), GP300 (an in frame deletion of ccpA), available in Stülke lab; WH649 (aphA3), available in Gerald Seidel's lab
- Expression vector:
- lacZ fusion:
- GFP fusion:
Labs working on this gene/protein
- Gerald Seidel, Erlangen University, Germany Homepage
- Richard Brennan, Houston, Texas, USA Homepage
- Milton H. Saier, University of California at San Diego, USA Homepage
- Yasutaro Fujita, University of Fukuyama, Japan
- Jörg Stülke, University of Göttingen, Germany Homepage
- Oscar Kuipers, University of Groningen, The Netherlands Homepage
Your additional remarks
References
Reviews
General and physiological studies
Additional publications: PubMed
Global analyses (proteome, transcriptome, ChIP-chip)
Bogumiła C Marciniak, Monika Pabijaniak, Anne de Jong, Robert Dűhring, Gerald Seidel, Wolfgang Hillen, Oscar P Kuipers
High- and low-affinity cre boxes for CcpA binding in Bacillus subtilis revealed by genome-wide analysis.
BMC Genomics: 2012, 13;401
[PubMed:22900538]
[WorldCat.org]
[DOI]
(I e)
Joerg Martin Buescher, Wolfram Liebermeister, Matthieu Jules, Markus Uhr, Jan Muntel, Eric Botella, Bernd Hessling, Roelco Jacobus Kleijn, Ludovic Le Chat, François Lecointe, Ulrike Mäder, Pierre Nicolas, Sjouke Piersma, Frank Rügheimer, Dörte Becher, Philippe Bessieres, Elena Bidnenko, Emma L Denham, Etienne Dervyn, Kevin M Devine, Geoff Doherty, Samuel Drulhe, Liza Felicori, Mark J Fogg, Anne Goelzer, Annette Hansen, Colin R Harwood, Michael Hecker, Sebastian Hubner, Claus Hultschig, Hanne Jarmer, Edda Klipp, Aurélie Leduc, Peter Lewis, Frank Molina, Philippe Noirot, Sabine Peres, Nathalie Pigeonneau, Susanne Pohl, Simon Rasmussen, Bernd Rinn, Marc Schaffer, Julian Schnidder, Benno Schwikowski, Jan Maarten Van Dijl, Patrick Veiga, Sean Walsh, Anthony J Wilkinson, Jörg Stelling, Stéphane Aymerich, Uwe Sauer
Global network reorganization during dynamic adaptations of Bacillus subtilis metabolism.
Science: 2012, 335(6072);1099-103
[PubMed:22383848]
[WorldCat.org]
[DOI]
(I p)
Andrzej T Lulko, Girbe Buist, Jan Kok, Oscar P Kuipers
Transcriptome analysis of temporal regulation of carbon metabolism by CcpA in Bacillus subtilis reveals additional target genes.
J Mol Microbiol Biotechnol: 2007, 12(1-2);82-95
[PubMed:17183215]
[WorldCat.org]
[DOI]
(P p)
Hans-Matti Blencke, Georg Homuth, Holger Ludwig, Ulrike Mäder, Michael Hecker, Jörg Stülke
Transcriptional profiling of gene expression in response to glucose in Bacillus subtilis: regulation of the central metabolic pathways.
Metab Eng: 2003, 5(2);133-49
[PubMed:12850135]
[WorldCat.org]
[DOI]
(P p)
M S Moreno, B L Schneider, R R Maile, W Weyler, M H Saier
Catabolite repression mediated by the CcpA protein in Bacillus subtilis: novel modes of regulation revealed by whole-genome analyses.
Mol Microbiol: 2001, 39(5);1366-81
[PubMed:11251851]
[WorldCat.org]
[DOI]
(P p)
K Yoshida, K Kobayashi, Y Miwa, C M Kang, M Matsunaga, H Yamaguchi, S Tojo, M Yamamoto, R Nishi, N Ogasawara, T Nakayama, Y Fujita
Combined transcriptome and proteome analysis as a powerful approach to study genes under glucose repression in Bacillus subtilis.
Nucleic Acids Res: 2001, 29(3);683-92
[PubMed:11160890]
[WorldCat.org]
[DOI]
(I p)
S Tobisch, D Zühlke, J Bernhardt, J Stülke, M Hecker
Role of CcpA in regulation of the central pathways of carbon catabolism in Bacillus subtilis.
J Bacteriol: 1999, 181(22);6996-7004
[PubMed:10559165]
[WorldCat.org]
[DOI]
(P p)
Repression of target genes by CcpA
Additional publications: PubMed
Positive regulation of gene expression by CcpA
Control of CcpA activity
CcpA-DNA interaction
Functional analysis of CcpA
H Ludwig, J Stülke
The Bacillus subtilis catabolite control protein CcpA exerts all its regulatory functions by DNA-binding.
FEMS Microbiol Lett: 2001, 203(1);125-9
[PubMed:11557150]
[WorldCat.org]
[DOI]
(P p)
E Küster-Schöck, A Wagner, U Völker, W Hillen
Mutations in catabolite control protein CcpA showing glucose-independent regulation in Bacillus megaterium.
J Bacteriol: 1999, 181(24);7634-8
[PubMed:10601226]
[WorldCat.org]
[DOI]
(P p)
E Küster, T Hilbich, M K Dahl, W Hillen
Mutations in catabolite control protein CcpA separating growth effects from catabolite repression.
J Bacteriol: 1999, 181(13);4125-8
[PubMed:10383986]
[WorldCat.org]
[DOI]
(P p)
A Kraus, E Küster, A Wagner, K Hoffmann, W Hillen
Identification of a co-repressor binding site in catabolite control protein CcpA.
Mol Microbiol: 1998, 30(5);955-63
[PubMed:9988473]
[WorldCat.org]
[DOI]
(P p)
A Kraus, W Hillen
Analysis of CcpA mutations defective in carbon catabolite repression in Bacillus megaterium.
FEMS Microbiol Lett: 1997, 153(1);221-6
[PubMed:9252590]
[WorldCat.org]
[DOI]
(P p)
Structural analyses
Bernhard Loll, Wolfram Saenger, Jacek Biesiadka
Structure of full-length transcription regulator CcpA in the apo form.
Biochim Biophys Acta: 2007, 1774(6);732-6
[PubMed:17500051]
[WorldCat.org]
[DOI]
(P p)
Rajesh Kumar Singh, Gottfried J Palm, Santosh Panjikar, Winfried Hinrichs
Structure of the apo form of the catabolite control protein A (CcpA) from Bacillus megaterium with a DNA-binding domain.
Acta Crystallogr Sect F Struct Biol Cryst Commun: 2007, 63(Pt 4);253-7
[PubMed:17401189]
[WorldCat.org]
[DOI]
(I p)
Maria A Schumacher, Gerald Seidel, Wolfgang Hillen, Richard G Brennan
Structural mechanism for the fine-tuning of CcpA function by the small molecule effectors glucose 6-phosphate and fructose 1,6-bisphosphate.
J Mol Biol: 2007, 368(4);1042-50
[PubMed:17376479]
[WorldCat.org]
[DOI]
(P p)
Vincent Chaptal, Virginie Gueguen-Chaignon, Sandrine Poncet, Cécile Lecampion, Philippe Meyer, Josef Deutscher, Anne Galinier, Sylvie Nessler, Solange Moréra
Structural analysis of B. subtilis CcpA effector binding site.
Proteins: 2006, 64(3);814-6
[PubMed:16755587]
[WorldCat.org]
[DOI]
(I p)
Maria A Schumacher, Gerald Seidel, Wolfgang Hillen, Richard G Brennan
Phosphoprotein Crh-Ser46-P displays altered binding to CcpA to effect carbon catabolite regulation.
J Biol Chem: 2006, 281(10);6793-800
[PubMed:16316990]
[WorldCat.org]
[DOI]
(P p)
Maria A Schumacher, Gregory S Allen, Marco Diel, Gerald Seidel, Wolfgang Hillen, Richard G Brennan
Structural basis for allosteric control of the transcription regulator CcpA by the phosphoprotein HPr-Ser46-P.
Cell: 2004, 118(6);731-41
[PubMed:15369672]
[WorldCat.org]
[DOI]
(P p)
J Tebbe, P Orth, E K Küster-Schöck, W Hillen, W Saenger, W Hinrichs
Crystallization and preliminary X-ray analyses of catabolite control protein A, free and in complex with its DNA-binding site.
Acta Crystallogr D Biol Crystallogr: 2000, 56(Pt 1);67-9
[PubMed:10666630]
[WorldCat.org]
[DOI]
(P p)