Difference between revisions of "PrsA"
| Line 1: | Line 1: | ||
| − | * '''Description:''' | + | * '''Description:''' post-translocational folding of exported proteins (post-translocation molecular chaperone) <br/><br/> |
{| align="right" border="1" cellpadding="2" | {| align="right" border="1" cellpadding="2" | ||
| Line 12: | Line 12: | ||
|style="background:#ABCDEF;" align="center"| '''Product''' || post-translocation molecular chaperone | |style="background:#ABCDEF;" align="center"| '''Product''' || post-translocation molecular chaperone | ||
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"|'''Function''' || protein | + | |style="background:#ABCDEF;" align="center"|'''Function''' || protein folding |
|- | |- | ||
|style="background:#ABCDEF;" align="center"| '''MW, pI''' || 32 kDa, 9.122 | |style="background:#ABCDEF;" align="center"| '''MW, pI''' || 32 kDa, 9.122 | ||
| Line 54: | Line 54: | ||
=== Basic information/ Evolution === | === Basic information/ Evolution === | ||
| − | * '''Catalyzed reaction/ biological activity:''' | + | * '''Catalyzed reaction/ biological activity:''' catalyses the post-translocational folding of exported proteins {{PubMed|20487272}}, required for folding of [[penicillin-binding proteins]] ([[PbpA]], [[PbpB]], [[PbpC]], [[PbpD]]) {{PubMed|20487272}} |
* '''Protein family:''' PpiC domain (according to Swiss-Prot) | * '''Protein family:''' PpiC domain (according to Swiss-Prot) | ||
| Line 72: | Line 72: | ||
* '''Effectors of protein activity:''' | * '''Effectors of protein activity:''' | ||
| − | * '''Interactions:''' | + | * '''Interactions:''' dimeric or oligomeric protein {{PubMed|20487272}} |
| − | * '''Localization:''' | + | * '''Localization:''' membrane associated {{PubMed|18763711,20487272}}, distinct spots organized in a helical pattern along the cell membrane {{PubMed|20487272}} |
=== Database entries === | === Database entries === | ||
| Line 120: | Line 120: | ||
=References= | =References= | ||
| − | <pubmed>12634326,14976191,11807061,10096076,18763711, 10871614 </pubmed> | + | <pubmed>12634326,14976191,11807061,10096076,18763711, 10871614 20487272 </pubmed> |
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 11:06, 22 May 2010
- Description: post-translocational folding of exported proteins (post-translocation molecular chaperone)
| Gene name | prsA |
| Synonyms | |
| Essential | yes PubMed |
| Product | post-translocation molecular chaperone |
| Function | protein folding |
| MW, pI | 32 kDa, 9.122 |
| Gene length, protein length | 876 bp, 292 aa |
| Immediate neighbours | yhaL, yhzE |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 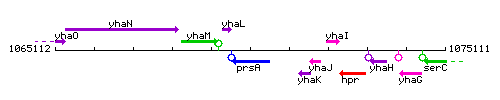
This image was kindly provided by SubtiList
| |
Contents
The gene
Basic information
- Locus tag: BSU09950
Phenotypes of a mutant
essential PubMed
Database entries
- DBTBS entry: no entry
- SubtiList entry: [1]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity: catalyses the post-translocational folding of exported proteins PubMed, required for folding of penicillin-binding proteins (PbpA, PbpB, PbpC, PbpD) PubMed
- Protein family: PpiC domain (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information:
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Interactions: dimeric or oligomeric protein PubMed
- Localization: membrane associated PubMed, distinct spots organized in a helical pattern along the cell membrane PubMed
Database entries
- Structure: 1ZK6
- UniProt: P24327
- KEGG entry: [2]
- E.C. number: 5.2.1.8
Additional information
Expression and regulation
- Operon:
- Regulation:
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References