Difference between revisions of "TsaE"
| Line 15: | Line 15: | ||
|colspan="2" style="background:#FAF8CC;" align="center"| '''Gene expression levels in [http://cellpublisher.gobics.de/subtiexpress/ ''Subti''Express]''': [http://cellpublisher.gobics.de/subtiexpress/bsu/BSU05910 ydiB] | |colspan="2" style="background:#FAF8CC;" align="center"| '''Gene expression levels in [http://cellpublisher.gobics.de/subtiexpress/ ''Subti''Express]''': [http://cellpublisher.gobics.de/subtiexpress/bsu/BSU05910 ydiB] | ||
|- | |- | ||
| − | |colspan="2" style="background:#FAF8CC;" align="center"| '''Interactions involving this protein in [http://cellpublisher.gobics.de/subtinteract/startpage/start/ ''Subt''Interact]''': [http://cellpublisher.gobics.de/subtinteract/interactionList/2/ | + | |colspan="2" style="background:#FAF8CC;" align="center"| '''Interactions involving this protein in [http://cellpublisher.gobics.de/subtinteract/startpage/start/ ''Subt''Interact]''': [http://cellpublisher.gobics.de/subtinteract/interactionList/2/TsaE TsaE] |
|- | |- | ||
|style="background:#ABCDEF;" align="center"| '''MW, pI''' || 17 kDa, 4.597 | |style="background:#ABCDEF;" align="center"| '''MW, pI''' || 17 kDa, 4.597 | ||
| Line 21: | Line 21: | ||
|style="background:#ABCDEF;" align="center"| '''Gene length, protein length''' || 474 bp, 158 aa | |style="background:#ABCDEF;" align="center"| '''Gene length, protein length''' || 474 bp, 158 aa | ||
|- | |- | ||
| − | |style="background:#ABCDEF;" align="center"|'''Immediate neighbours''' || ''[[thiL]]'', ''[[ | + | |style="background:#ABCDEF;" align="center"|'''Immediate neighbours''' || ''[[thiL]]'', ''[[tsaB]]'' |
|- | |- | ||
|colspan="2" style="background:#FAF8CC;" align="center"|'''Get the DNA and protein [http://srs.ebi.ac.uk/srsbin/cgi-bin/wgetz?-e+[EMBLCDS:CAB12410]+-newId sequences] <br/> (Barbe ''et al.'', 2009)''' | |colspan="2" style="background:#FAF8CC;" align="center"|'''Get the DNA and protein [http://srs.ebi.ac.uk/srsbin/cgi-bin/wgetz?-e+[EMBLCDS:CAB12410]+-newId sequences] <br/> (Barbe ''et al.'', 2009)''' | ||
| Line 94: | Line 94: | ||
* '''[[Localization]]:''' | * '''[[Localization]]:''' | ||
| − | ** cytoplasm with the | + | ** cytoplasm with the exception of the nucleoid region [http://www.ncbi.nlm.nih.gov/sites/entrez/19246765 PubMed] |
=== Database entries === | === Database entries === | ||
| Line 112: | Line 112: | ||
=Expression and regulation= | =Expression and regulation= | ||
| − | * '''Operon:''' | + | * '''Operon:''' ''[[thiL]]-[[tsaE]]-[[tsaB]]-[[ydiD]]-[[tsaD]]'' {{PubMed|22383849}} |
| − | * '''Expression browser:''' [http://genome.jouy.inra.fr/cgi-bin/seb/viewdetail.py?id=ydiB_641654_642130_1 | + | * '''Expression browser:''' [http://genome.jouy.inra.fr/cgi-bin/seb/viewdetail.py?id=ydiB_641654_642130_1 tsaE] {{PubMed|22383849}} |
* '''Sigma factor:''' | * '''Sigma factor:''' | ||
| Line 146: | Line 146: | ||
=References= | =References= | ||
| − | <pubmed> 17005971, 19246765 23072323</pubmed> | + | <pubmed> 17005971, 19246765 23072323 22383849</pubmed> |
[[Category:Protein-coding genes]] | [[Category:Protein-coding genes]] | ||
Revision as of 12:24, 18 October 2012
- Description: P-loop ATPase, required for threonyl carbamoyl adenosine (t6A) modification of tRNAs that pair with ANN codons in mRNA
| Gene name | ydiB |
| Synonyms | |
| Essential | no |
| Product | tRNA modification enzyme |
| Function | tRNA modification |
| Gene expression levels in SubtiExpress: ydiB | |
| Interactions involving this protein in SubtInteract: TsaE | |
| MW, pI | 17 kDa, 4.597 |
| Gene length, protein length | 474 bp, 158 aa |
| Immediate neighbours | thiL, tsaB |
| Get the DNA and protein sequences (Barbe et al., 2009) | |
Genetic context 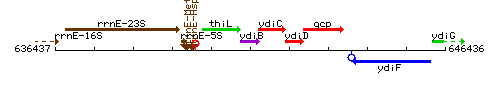
This image was kindly provided by SubtiList
| |
Expression at a glance PubMed
| |
Contents
Categories containing this gene/protein
This gene is a member of the following regulons
The gene
Basic information
- Locus tag: BSU05910
Phenotypes of a mutant
reduced growth rate PubMed
Database entries
- DBTBS entry: no entry
- SubtiList entry: [1]
Additional information
The protein
Basic information/ Evolution
- Catalyzed reaction/ biological activity:
- Protein family: UPF0079 family (according to Swiss-Prot)
- Paralogous protein(s):
Extended information on the protein
- Kinetic information: K(M) for ATP: 60 myM, V(max) 10 nmol/min PubMed
- Domains:
- Modification:
- Cofactor(s):
- Effectors of protein activity:
- Localization:
- cytoplasm with the exception of the nucleoid region PubMed
Database entries
- Structure:
- UniProt: O05515
- KEGG entry: [2]
- E.C. number:
Additional information
ATPase activity is required for in vivo function of the protein PubMed
Expression and regulation
- Sigma factor:
- Regulation:
- Regulatory mechanism:
- Additional information:
Biological materials
- Mutant:
- Expression vector:
- lacZ fusion:
- GFP fusion:
- two-hybrid system:
- Antibody:
Labs working on this gene/protein
Your additional remarks
References